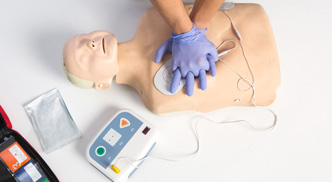
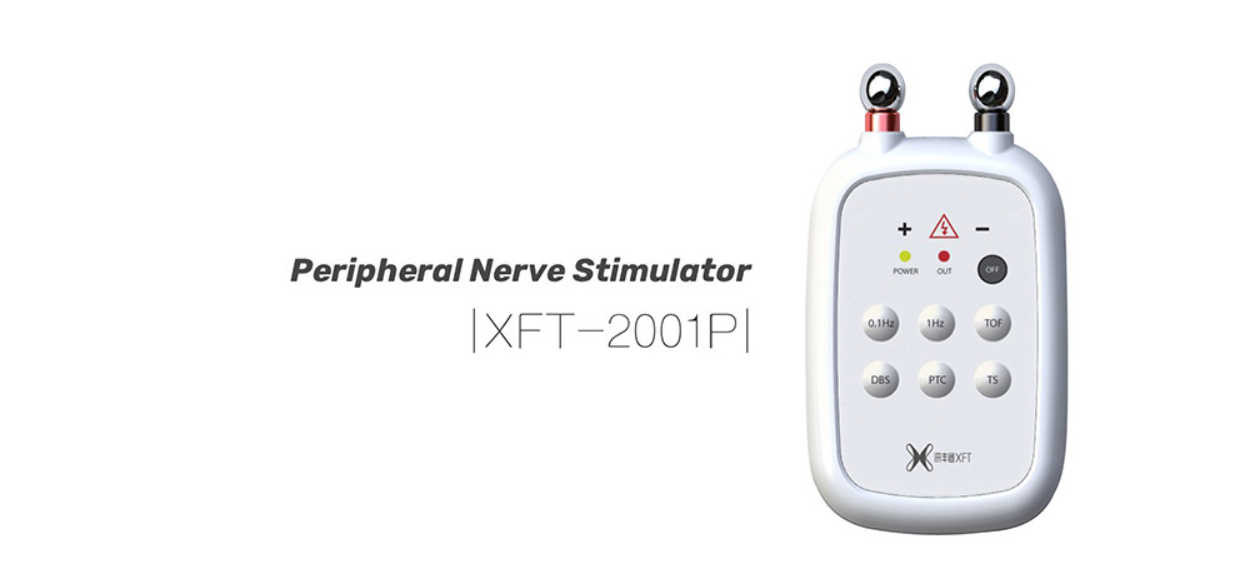
Foot Drop


Book a Solution
Book a Solution


About XFT
Technology Upgrades Our Life
XFT headquartered in Shenzhen National Bio-Industrial Park , with 10,000 square meters of modern production research and development center in line with the GMP standards.

Rehabilitation
Technology Upgrades Our Life
XFT is committed to neurological rehabilitation solutions, including bio-feedback, NMES, mirror therapy, HCI technology.